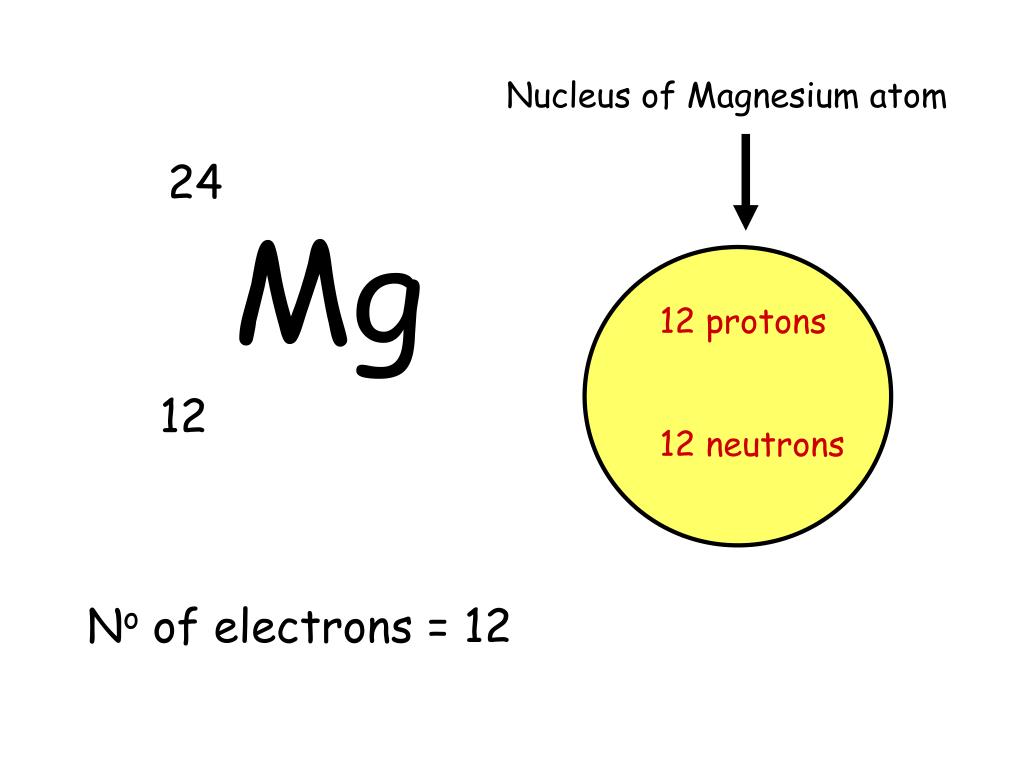
Magnesium Nuclear Charge . Subtract this value from the nuclear. what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. But looking at magnesium which has 10. First we must determine the electron configuration of magnesium to determine the number of. approximate the effective nuclear charge of magnesium. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. First, compute the overall shielding effect of the electrons orbiting the nucleus. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. to calculate the effective nuclear charge:
from www.slideserve.com
But looking at magnesium which has 10. First, compute the overall shielding effect of the electrons orbiting the nucleus. to calculate the effective nuclear charge: approximate the effective nuclear charge of magnesium. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. First we must determine the electron configuration of magnesium to determine the number of. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. Subtract this value from the nuclear.
PPT Nuclear model of atom PowerPoint Presentation, free download ID
Magnesium Nuclear Charge charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. Subtract this value from the nuclear. But looking at magnesium which has 10. First, compute the overall shielding effect of the electrons orbiting the nucleus. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. First we must determine the electron configuration of magnesium to determine the number of. approximate the effective nuclear charge of magnesium. to calculate the effective nuclear charge:
From www.alamy.com
Magnesium (Mg). Diagram of the nuclear composition and electron Magnesium Nuclear Charge But looking at magnesium which has 10. First, compute the overall shielding effect of the electrons orbiting the nucleus. First we must determine the electron configuration of magnesium to determine the number of. approximate the effective nuclear charge of magnesium. to calculate the effective nuclear charge: Subtract this value from the nuclear. what is the effective attraction. Magnesium Nuclear Charge.
From www.alamy.com
Symbol and electron diagram for Magnesium illustration Stock Vector Magnesium Nuclear Charge approximate the effective nuclear charge of magnesium. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. First we must determine the electron configuration of magnesium to determine the number of. what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the. Magnesium Nuclear Charge.
From www.alamy.com
magnesium isotopes atomic structure backdrop physics theory Magnesium Nuclear Charge to calculate the effective nuclear charge: First, compute the overall shielding effect of the electrons orbiting the nucleus. what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. Subtract this value from the nuclear. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution. Magnesium Nuclear Charge.
From enginedatanichered.z21.web.core.windows.net
Atomic Diagram Of Magnesium Magnesium Nuclear Charge the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. First we must determine the electron configuration of magnesium to determine the number of. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. Subtract. Magnesium Nuclear Charge.
From enginedatanichered.z21.web.core.windows.net
Magnesium Atom Diagram Magnesium Nuclear Charge what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. to calculate the effective nuclear charge: approximate the effective nuclear charge of magnesium. First we must determine the. Magnesium Nuclear Charge.
From www.webelements.com
Elements Periodic Table » Magnesium » properties of free atoms Magnesium Nuclear Charge First we must determine the electron configuration of magnesium to determine the number of. But looking at magnesium which has 10. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. First, compute the overall shielding effect of the electrons orbiting the nucleus. approximate the. Magnesium Nuclear Charge.
From commons.wikimedia.org
FileElectron shell 012 magnesium.png Wikimedia Commons Magnesium Nuclear Charge what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. approximate the effective nuclear charge of magnesium. Subtract this value from the nuclear. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. First we must. Magnesium Nuclear Charge.
From valenceelectrons.com
Magnesium Electron Configuration Aufbau & Bohr Model Magnesium Nuclear Charge First we must determine the electron configuration of magnesium to determine the number of. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. to calculate. Magnesium Nuclear Charge.
From www.alamy.com
3d render of atom structure of magnesium isolated over white background Magnesium Nuclear Charge what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. approximate the effective nuclear charge of magnesium. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. to calculate the effective nuclear charge: charge. Magnesium Nuclear Charge.
From enginelistjuprefecture.z21.web.core.windows.net
Magnesium Atomic Structure Diagram Magnesium Nuclear Charge what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. to calculate the effective nuclear charge: First, compute the overall shielding effect of the electrons orbiting the nucleus. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons. Magnesium Nuclear Charge.
From www.youtube.com
Effective Nuclear Charge Chemistry Tutorial YouTube Magnesium Nuclear Charge approximate the effective nuclear charge of magnesium. First, compute the overall shielding effect of the electrons orbiting the nucleus. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. First we must determine the electron configuration of magnesium to determine the number of. what. Magnesium Nuclear Charge.
From www.chegg.com
Solved Calculate the effective nuclear charge experienced by Magnesium Nuclear Charge But looking at magnesium which has 10. the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. Subtract this value from the nuclear. First, compute the overall shielding effect of the electrons orbiting the nucleus. First we must determine the electron configuration of magnesium to determine. Magnesium Nuclear Charge.
From userdatadorgan.z21.web.core.windows.net
Magnesium Aluminum Phase Diagram Magnesium Nuclear Charge what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. Subtract this value from the nuclear. But looking at magnesium which has 10. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. First we must determine the electron configuration. Magnesium Nuclear Charge.
From www.wizeprep.com
Effective Nuclear Charge (Zeff) Wize University Chemistry Textbook Magnesium Nuclear Charge the effective nuclear charge changes relatively little for electrons in the outermost, or valence shell, from lithium to cesium because electrons in filled. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. Subtract this value from the nuclear. to calculate the effective nuclear charge: But looking. Magnesium Nuclear Charge.
From www.alamy.com
magnesium isotopes atomic structure backdrop physics theory Magnesium Nuclear Charge charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. But looking at magnesium which has 10. First, compute the overall shielding effect of the electrons orbiting the nucleus. First we must determine the electron configuration of magnesium to determine the number of. Subtract this value from the nuclear.. Magnesium Nuclear Charge.
From sites.google.com
Atomic Structure Protons, Neutrons and Electrons Mrs. Sanborn's Site Magnesium Nuclear Charge to calculate the effective nuclear charge: First we must determine the electron configuration of magnesium to determine the number of. Subtract this value from the nuclear. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution of the nuclear shape throughout. First, compute the overall shielding effect of the electrons orbiting the nucleus.. Magnesium Nuclear Charge.
From cejxrkef.blob.core.windows.net
Magnesium Ion Nuclear Notation at Catherine Lackey blog Magnesium Nuclear Charge Subtract this value from the nuclear. First, compute the overall shielding effect of the electrons orbiting the nucleus. what is the effective attraction \(z_{eff}\) experienced by the valence electrons in the magnesium anion, the neutral magnesium. But looking at magnesium which has 10. charge radii of all magnesium isotopes in the sd shell have been measured, revealing evolution. Magnesium Nuclear Charge.
From www.slideserve.com
PPT The Octet Rule PowerPoint Presentation, free download ID2654700 Magnesium Nuclear Charge First we must determine the electron configuration of magnesium to determine the number of. approximate the effective nuclear charge of magnesium. Subtract this value from the nuclear. But looking at magnesium which has 10. First, compute the overall shielding effect of the electrons orbiting the nucleus. charge radii of all magnesium isotopes in the sd shell have been. Magnesium Nuclear Charge.